What is meant by the term "Death, per Protocol" in the Brazil Fluvoxamine trial (NCT04727424) that was referenced by Steve Kirsch and funded by the Bill and Melinda Gates Foundation?
Steve Kirsch published Fluvoxamine for COVID: what you need to know, on November 05, 2021.
Below is an excerpt from Kirsch’s referencing NCT04727424.
Steve Kirsch: The evidence is solid. There are 4 outpatient studies that have been done (2 at WashU (see Phase 2 trial results published in JAMA), one in Berkeley, CA by David Seftel, one in Brazil published in the Lancet, and one in-patient study done in Croatia. Three of the four outpatient trials have been reported out: all were successful. The WashU Phase 3 study hasn’t been disclosed yet, but they had compliance problems with their patients this time around (phase 2 was local so the patients got the drug early and also were very compliant and the placebo group was truly taking nothing). There were no studies reported out so far where fluvoxamine made things worse or neutral. All the supporting observational studies were positive as well.
The “one in Brazil published in Lancent” that Kirsch referred to links to this paper: Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial.
This paper in turn references trial #NCT04727424.
The trial is registered at ClinicalTrials.gov (NCT04727424) and is ongoing.
Although the trials began back in 2020, and they were “stopped” on August 5, 2021, according to the study authors, clinicaltrials.gov says that “no results have been posted” for NCT04727424 yet.
The Lancent paper did report results. Some other information from the paper:
The study team screened 9803 potential participants for this trial. The trial was initiated on June 2, 2020, with the current protocol reporting randomisation to fluvoxamine from Jan 20 to Aug 5, 2021, when the trial arms were stopped for superiority. 741 patients were allocated to fluvoxamine and 756 to placebo..
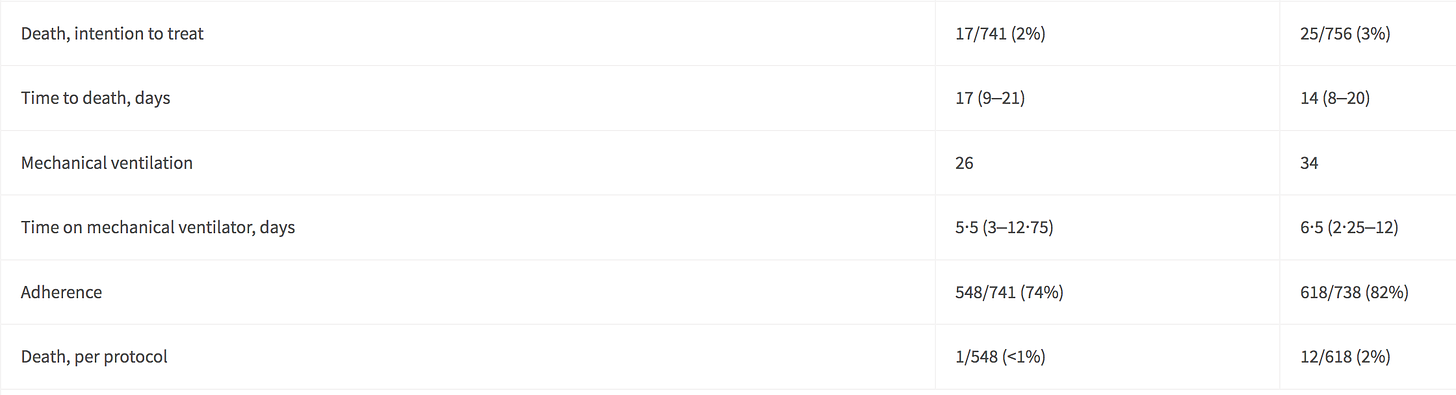
Here are the figures on Death.
Table 3. Secondary outcomes of fluvoxamine versus placebo in the TOGETHER trial
A summary of Death:
In the Fluvoxamine group, 26 out of 741 were mechanically ventilated.
In the Placebo group, 34 out of 756 were mechanically ventilated.
In the Fluvoxamine group, 17 died “with intention to treat.” 1 died “per protocol.”
In the Placebo Group, 25 died “with intention to treat.” 12 died “per protocol.”
I do not understand why death would be divided into two categories: A), with intention to treat, and B), per protocol.
I do not understand why “Death, per protocol” would be drastically higher in the Placebo group than the Fluvoxamine group. Death “with intent to treat” was 34 to 26, but “Death, per protocol” was 12 to 1 in the Fluvoxamine and Placebo groups.
There is some language in the Lancent paper on the protocol, but it really makes no sense to me. I won’t try to interpret it. The paper says:
The full protocol and statistical analysis plan have previously been published,9 and additional details are in appendix 2 (p 2).
The superscript 9 references this this link to Gates Open Research: https://gatesopenresearch.org/articles/5-117/v1.
The website for Gates Open Research states:
Gates Open Research is a platform for rapid author-led publication and open peer review of research funded by the Bill & Melinda Gates Foundation.
Here is the website page referenced to in the Lancent paper describing study details 9 for the Fluvoxamine Brazil trials:
The TOGETHER Trial is supported by the Bill and Melinda Gates Foundation [INV-019641]. Overall trial infrastructure is also supported by the Rainwater Foundation, with additional funding support from FastGrants for current evaluations of trial interventions. Future evaluations of interventions may be supported by other funding organizations. The funders have no role in the conduct, interpretation, or dissemination of the study findings.
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Interestingly, the Lancent paper lists the Rainwater Charitable Foundation and FastGrants as providing “Funding,” but not the Bill and Melinda Gates Foundation.
Funding
FastGrants and The Rainwater Charitable Foundation.
Gates Open Research, on the other hand, lists Rainwater and FastGrants as providing “overall trial infrastructure.”
As with Gates Open Research, the Lancent paper stated:
“The funders of the study had no role in the study design, data collection, data analysis, data interpretation, writing of the report, or decision to submit for publication.”
Aside: Perhaps the authors of the Lancent paper were reluctant to mention the Bill and Melinda Gates Foundation as a Funder of their study due to the Foundation’s reputation as an organization devoted to reducing global population with poisons disguised as Medicine.
Steve Kirsch and Bill Gates know each other. Kirsch wrote that:
Bill Gates is super impressive. I’ve known him for over 40 years.
Steve Kirsch defends the motivatios of Bill Gates, as he did to Sean Stone on February 1, 2023. In this interview with Stone, Kirsch also openly supported “Malthusian” goals of limiting population. (Thomas Malthus ((1766-1834) was a British Economist who stated that population should be controlled). Nevertheless, population continued to grow far beyond the estimates of Malthus. People today jokingly say today that there are “7 Billion reasons why Thomas Malthus was wrong.”
Like Kirsch, Bill Gates is a Malthusian. 2018 Bill Gates referred to Malthusian theory while pointing at the chart of population growth proving that Malthus was wrong. As Kirsch did, Gates said that people choose to have less children as they become healthier. Others say that vaccines have been used to sterilize women against their will.
That ends the aside.
SUMMARY:
It appears very possible the people who administered the Fluvoxamide trials in Brazil may have placed their hands on the scale to acheive a lower mortality rate of Fluvoxamine in comparison to the Placebo group. Of course I am aware that this is a strong allegation.
If the study authors and trials personnel wish to clear their name of this, they can do a few things. They can publish all relevant documents that pertain to the said “protocol.” They can provide verbal descriptions on how the protocol was actually administered in the trials. They can explain why they think the rate of “death, per protocol” was so much higher in the Placebo group.
They can list the doctor or doctors and staff who attended each death. Perhaps some doctors and staff were more likely to have high mortality rates than others.
This is the kind of data the public really needs, Mr. Kirsch. Perhaps you could help? Perhaps this is study is a “smoking gun,” as you describe to Sean Stone?
Charles Wright
P.S. If you wish to report my research please consider becoming a paid subscriber, or “buy me a coffee.”




As a Brazilian, I STRONGLY RECOMEND that nobody take seriously, any so-called medical research done at any Brazilian university.
I know Malone has acknowledged psychiatric problems, at least in the past, but hasn't Kirsch mentioned having ADHD or something else? I could be very wrong about Kirsch. Many times people who take those drugs fall in love with them, and become huge advocates. I think that explains the Malone lawsuit against the Breggins, and Kirsch trying to normalize the taking of an SSRI.